Ashraf Engineer
April 17, 2021
Hello and welcome to All Indians Matter. I am Ashraf Engineer.
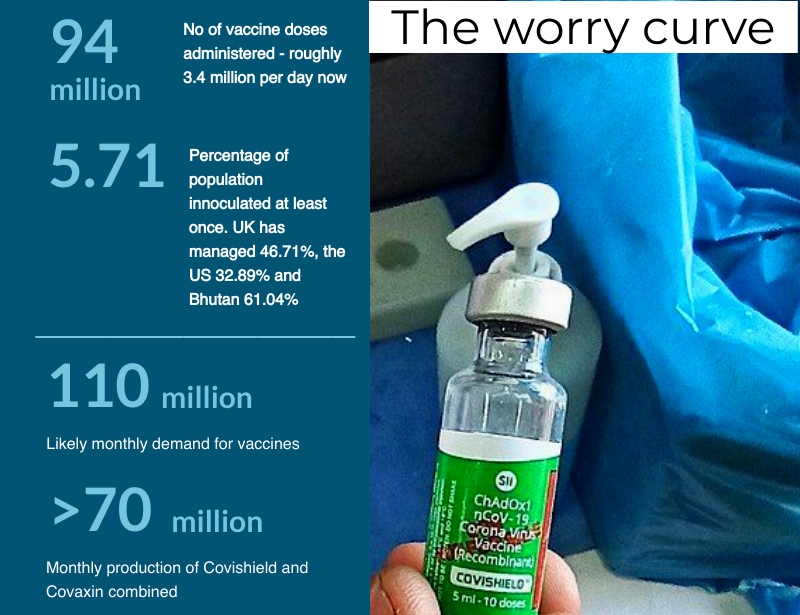
We are well and truly in the midst of the second wave of COVID-19. From roughly 11,000 new cases every day in February, India was registering 131,000 by the first week of April and 184,000 on April 13. The fear is that we haven’t hit the peak yet. Naturally, India’s preparedness is in question. India is a vaccine manufacturing powerhouse but, ironically, it suffers from a severe vaccine shortage. At the time of making this podcast, India had administered 94 million doses with daily administration at 3.4 million. The vaccine scarcity has been compounded by a shortage of Remdesivir, the injectable anti-viral formulated to treat Ebola but repurposed in 2020 to treat COVID-19. It’s a double whammy that has implications not just for public health but for the economy too.
SIGNATURE TUNE
The second wave of COVID-19 has put health workers, policymakers and the government on edge. The numbers are worrying and the massive rallies held for recent state elections as well as the Maha Kumbh at which lakhs gathered in Haridwar with little adherence to social distancing or mask use will not help. Those infected at these events will go on to infect others, increasing the case load. It’s not looking good.
Let’s return to the vaccine. The number of doses administered may look large but, taken as a percentage of the population, India doesn’t fare well. According to data tracking portal OurWorldinData, India has vaccinated 5.71% of its population at least once. The UK, meanwhile, has managed 46.71% and the US 32.89%. Even Bhutan with 61.04% and Brazil at 8.87% have done better. The problem, of course, is our large population that makes even the big numbers seem small.
India started off by allowing two vaccines — Covishield and Covaxin. Covishield is manufactured by the Serum Institute of India and is our version of the vaccine developed by Astra Zeneca and Oxford University. It is simpler to store and transport, and is cheaper to make.
Covaxin is developed by a consortium that includes the Indian Council for Medical Research and is manufactured by Bharat Biotech. There were some doubts about it initially but it has done well in Phase 2 trials.
India is using Covishield mainly with the Serum Institute supplying more than 100 million doses to the government and exporting 60 million. Its production capacity is 60 million per month while less than 10 million doses of Covaxin are produced every month.
Here’s the problem: with the second wave upon us, the demand is likely to be 3.5 million doses per day or just under 110 million doses per month. Covishield and Covaxin combined give us less than 70 million.
As a result, the government has been forced to give emergency approval to vaccines made by foreign manufacturers – essentially those already approved by the US, UK and Japan, and those listed by the World Health Organization. Among these is the much talked about Sputnik V from Russia.
Now, this emergency approval is good and bad. It will mean more doses available in India but it also means a major relaxation of testing. As per the New Drugs and Clinical Trials Rules, 2019, foreign manufacturers applying for emergency authorisation must submit the results of local clinical trials, which are called bridging trials.
But the rules also allow the relaxation of norms if the vaccine has been approved by another country. This is the clause that has been invoked to allow foreign vaccines into India – we have waived the condition to conduct bridging trials at Indian sites.
India has, however, introduced a special condition – foreign manufacturers must assess the vaccine’s impact on the first 100 beneficiaries for seven days before rolling it out for wider administration.
Let’s talk about Remdesivir now. As demand surged, huge crowds gathered outside hospitals wanting to buy the drug for family members but were turned away as stocks ran out. The government responded by banning its export.
The clamour for Remdesivir is an indication of how desperate people are for treatment. The World Health Organization has said there’s no evidence that the drug improves survival and other outcomes but, in the absence of anything else, people are turning to it, hoping against hope.
When the infection curve flattened after the first wave, Indian firms cut production of Remdesivir. However, the aggressive resurgence has led to hoarding, over-prescription and shortages.
Whether it’s a vaccine shortage or that of Remdesivir, it’s clear that India isn’t prepared. When case numbers slid in India, Europe and the US were already in the midst of second and third waves. So, the Indian government had fair warning. The government did not, however, offer enough financial help to vaccine makers or sign purchase orders or pay up in advance. This hampered capacity expansion.
As things stand, the numbers are more worrying now than when the pandemic first hit and lockdowns of various intensities have been reimposed in several states. Maharashtra, perhaps the state most crucial to India’s economy, has imposed a limited lockdown and several other states are clocking a high number of cases.
Once again, bereft migrant labourers are returning to their villages and the curbs will most certainly impact economic activity.
Lack of planning is in danger of erasing any gains India might have made in tackling the pandemic. The curve is rising fast. Can the government move faster?
Thank you for listening. Please visit allindiansmatter.in for more columns and audio podcasts. You can follow me on Twitter at @AshrafEngineer and @AllIndiansCount. Search for the All Indians Matter page on Facebook. On Instagram, the handle is @AllIndiansMatter. Email me at editor@www.allindiansmatter.in. Catch you again soon.